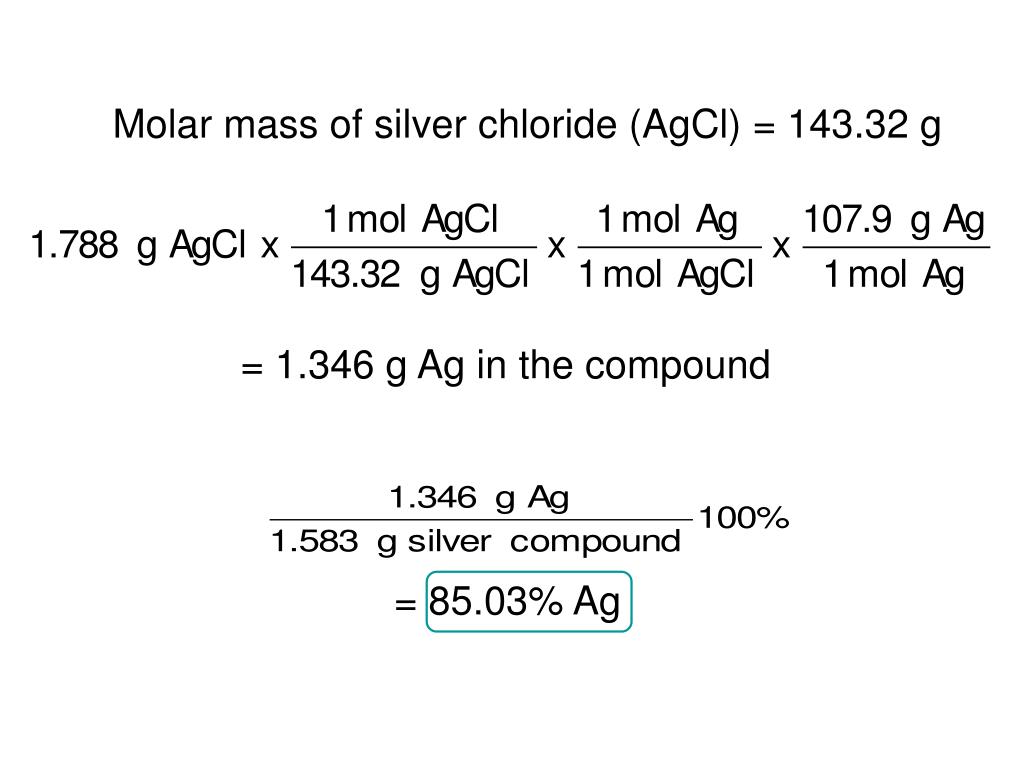
So GaBr 3 accounts for 29.6% of the mass of the unknown solid. So there were 0.202g GaBr 3 originally present in the solid mixture. There are at least four high pressure forms: -MgH2 with -PbO2 structure, cubic -MgH2 with Pa-3 space group, orthorhombic HP1 with Pbc21 space group and orthorhombic HP2 with Pnma space group. Molar mass GaBr 3 = 69.7 + 3(79.9) = 309.4g/mol GaBr 3Ġ.000653 mol GaBr 3 x (309.4g/1 mol GaBr 3) = 0.202g GaBr 3 The room temperature form -MgH2 has a rutile structure. We want to know what percent of the original 0.6813g of solid is GaBr 3 ? So we must convert moles of GaBr 3 to grams of GaBr 3.Īgain, the molar mass presents itself for the finding.I trust you.I have faith in you. So what is the question? Let's not lose sight of that. So we must have started with 0.000653 mol GaBr 3Īlmost there peeps. So we set up a unit conversion.Ġ.00196 mol AgBr x (1 mol GaBr 3/3 mol AgBr) = 0.000653 mol GaBr 3 This equation gives us a 1 to 3 ratio of GaBr 3 to AgBr. Now we ask ourselves, how can we relate moles of AgBr to moles of GaBr 3? The stoichiometric ratio. So to find the molar mass of AgBr (you may be way ahead of me here), we are going to add them together. Referring to the periodic table, the molar mass of Ag is 107.9g/mol, and the molar mass of Br is 79.9g/mol. So let's find out how many moles of the product were made. If we know how many moles of a product we have, we can use the stoichiometric ratio to find out how many moles of reactant we have. If we know how many grams of product were produced, then we can determine how many moles were produced (via the molar mass). And we know how many grams of one product is produced. In particular, hydrogen bonds are responsible for the fact that water is a liquid at temperatures at which molecules of similar molecular mass are gases. Now what's the deal with this excess AgNO 3? Well according to what they tell us, AgNO 3 reacts with GaBr 3, producing two products. 2, in the solution, in mol dm3 Molar mass of Fe2(SO4)3H2O 417.9 g mol1. What this question boils down to is this: What is the mass of GaBr 3 in this solid mixture? (Forget what else is in there.who even cares.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
